- Blog
- Kana kanum kalangal kallooriyin kadhai 2012
- Where to watch naruto online english dub free reddit
- Archicad 18 google earth
- Marshall avt 50h 2000
- Realtek hd audio manager wont open
- Jennifer lopez u turn 4k download
- Suite life of zack and cody season 3 episode 11
- Lonewolf37 house flipper game
- How to use vst syntorial
- Ilk kuran tefsiri
- Ps2 game saves to ps3
- Download advanced rar repair
- Download kumpulan dlc mafia 2 pc
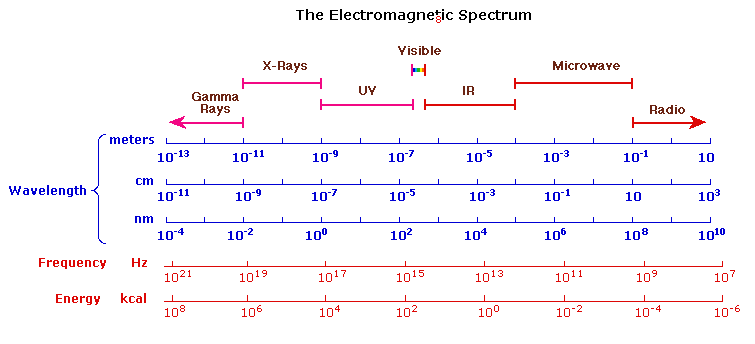
- Cut off wavelength solvent

These orbitals are absent in typical alkenes such as ethylene Carbonyl groups contain non-bonding electrons that are in an orbital intermediate in energy between the bonding pi orbital and the anti bonding pi* orbital.Huh? Let’s look at a simple molecular orbital drawing of acetone. Carbonyl (C=O) Groups Tend To Show Weak Absorbances At (Roughly) 300 nm That Correspond To Transitions Between Non-Bonding Orbitals and Pi* Orbitals It’s a transition from a non-bonding orbital (n) to the pi* orbital (n→π*).
Now: as we’ll see in a minute, there is a pi to pi* ( π→π*) transition for acetone in the UV, but that peak at 275 nm is NOT a pi to pi* transition. Wouldn’t you reasonably expect *more* energy to be required to promote an electron from pi(π) to pi* (π*)? If anything, C=O π bonds are stronger than C=C π bonds. If you have an astonishingly good memory you may recall from the last post (or from my introduction above) that the absorption max for ethene (CH 2=CH 2) is about 170 nm.Īn absorption around 275 nm means that longer wavelength and therefore less energetic photons are required for this transition. Here’s the UV-Vis absorption spectrum for 2-propanone (acetone). Question: Does acetone absorb UV or visible light?Īnswer: You betcha. Let’s start with one of the simplest compounds with a C=O bond: 2-propanone, otherwise known as acetone. Absorbance of C=O bonds Show A Maximum Around 300 nm

In our last postwe showed that molecules with C-C pi (π) bonds absorb light in the UV-visible region, which promotes electrons from (bonding) π orbitals to (anti bonding) π* orbitals. A Quick Review Of What We’ve Learned So Far About UV-Vis Summary: UV-Vis Spectroscopy Of Carbonylsġ.Carbonyls also participate in conjugation.This is actually a n-> pi* transition, not pi to pi* (!).C=O Bonds Show An Absorbance Maximum Around 300 nm.UV-Vis Spectroscopy Of Carbonyls (C=O Bonds)
- Blog
- Kana kanum kalangal kallooriyin kadhai 2012
- Where to watch naruto online english dub free reddit
- Archicad 18 google earth
- Marshall avt 50h 2000
- Realtek hd audio manager wont open
- Jennifer lopez u turn 4k download
- Suite life of zack and cody season 3 episode 11
- Lonewolf37 house flipper game
- How to use vst syntorial
- Ilk kuran tefsiri
- Ps2 game saves to ps3
- Download advanced rar repair
- Download kumpulan dlc mafia 2 pc
- Cut off wavelength solvent
